China's Refractories ›› 2022, Vol. 31 ›› Issue (1): 16-23.DOI: 10.19691/j.cnki.1004-4493.2022.01.003
Previous Articles Next Articles
Sintering Complexity of Magnesia-chrome Refractories
GUO Zongqi1,*( ), MA Ying2, LI Yong3
), MA Ying2, LI Yong3
- 1 School of Materials Science and Engineering, Xi’an University of Architecture and Technology,Xi’an 710055, China
2 RHI Magnesita (Dalian) Co., Ltd., Dalian 116600, China
3 School of Materials Science and Engineering, University of Science and Technology Beijing,Beijing 100083, China
-
Online:2022-03-15Published:2022-04-02 -
Contact:GUO Zongqi -
About author:Dr. Guo Zongqi studied the major of refractories in Xi’an University of Architecture and Technology, China, and received his PhD degree in Ecole Polytechnique, University of Montreal, Canada. For more than 10 years, he has been working on the syntheses of chromia raw materials and pioneering development of high chrome refractories for slagging coal gasifiers, which was awarded with Second Grade Technical Advance Prize by the Ministry of Chemical Industry. Dr. Guo has gained broad research experience on a few of international refractory platforms in Canada, Austria, Poland and China. His recent investigations and industrial practice include a great progress of high purity, high density sintered magnesia from natural magnesite. He has been involving in long-term continuous studies of burnt and unburnt basic bricks for cement rotary kilns, glass-making regenerators and steel-making process.
Cite this article
GUO Zongqi, MA Ying, LI Yong. Sintering Complexity of Magnesia-chrome Refractories[J]. China's Refractories, 2022, 31(1): 16-23.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://www.cnref.cn/EN/10.19691/j.cnki.1004-4493.2022.01.003
| Catalogue | Type I | Type II | Type III |
|---|---|---|---|
| Bonding mode | Silicate-bonded magnesia-chrome | Direct-bonded magnesia-chrome | Rebonded fused-grain magnesia-chrome |
| Aggregate as main component | Mid-grade sintered magnesia | High grade magnesia | Fused magnesia chrome |
| Chemical composition /mass% | |||
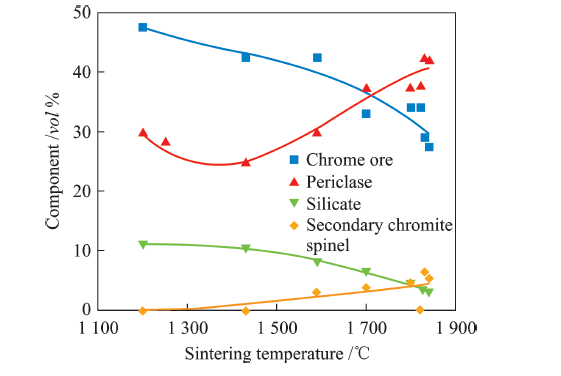
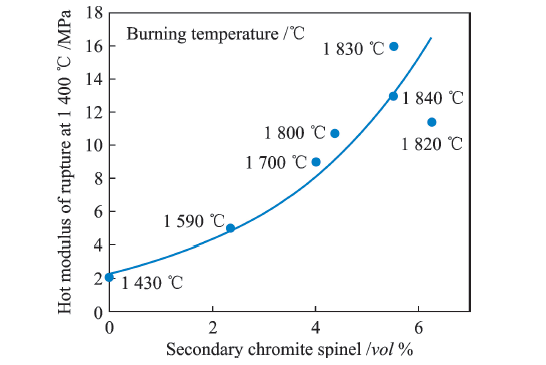
| MgO | 61.12 | 58.90 | 58.45 |
| Cr2O3 | 18.89 | 20.80 | 20.89 |
| Al2O3 | 5.86 | 6.66 | 6.73 |
| Fe2O3 | 11.20 | 12.50 | 12.28 |
| CaO | 1.04 | 0.84 | 0.68 |
| SiO2 | 1.31 | 0.43 | 0.56 |
| Bulk density /(g · cm-3) | 3.20 | 3.27 | 3.31 |
| Apparent porosity /% | 16.5 | 15.5 | 15.0 |
| Cold crushing strength /MPa | 60.0 | 62.5 | 70.0 |
| Thermal expansion at 1 600 °C /% | 1.8 | 1.7 | 1.7 |
| Hot modulus of rupture /MPa | |||
| at 1 250 °C | 17.2 | 14.0 | |
| at 1 400 °C | 3.4 | 5.2 | 9.2 |
| at 1 500 °C | 1.2 | 3.1 | 6.4 |
Table 1 Typical catalogues of commercial magnesia-chrome bricks
| Catalogue | Type I | Type II | Type III |
|---|---|---|---|
| Bonding mode | Silicate-bonded magnesia-chrome | Direct-bonded magnesia-chrome | Rebonded fused-grain magnesia-chrome |
| Aggregate as main component | Mid-grade sintered magnesia | High grade magnesia | Fused magnesia chrome |
| Chemical composition /mass% | |||
| MgO | 61.12 | 58.90 | 58.45 |
| Cr2O3 | 18.89 | 20.80 | 20.89 |
| Al2O3 | 5.86 | 6.66 | 6.73 |
| Fe2O3 | 11.20 | 12.50 | 12.28 |
| CaO | 1.04 | 0.84 | 0.68 |
| SiO2 | 1.31 | 0.43 | 0.56 |
| Bulk density /(g · cm-3) | 3.20 | 3.27 | 3.31 |
| Apparent porosity /% | 16.5 | 15.5 | 15.0 |
| Cold crushing strength /MPa | 60.0 | 62.5 | 70.0 |
| Thermal expansion at 1 600 °C /% | 1.8 | 1.7 | 1.7 |
| Hot modulus of rupture /MPa | |||
| at 1 250 °C | 17.2 | 14.0 | |
| at 1 400 °C | 3.4 | 5.2 | 9.2 |
| at 1 500 °C | 1.2 | 3.1 | 6.4 |
| Source of chrome ore | South Africa | India |
|---|---|---|
| SiO2 | 0.31 | 0.83 |
| Al2O3 | 14.66 | 10.54 |
| Fe2O3 | 27.87 | 16.71 |
| CaO | 0 | 0.36 |
| MgO | 10.28 | 12.67 |
| Cr2O3 | 46.12 | 58.71 |
| LOI | 0.76 | 0.18 |
Table 2 Chemical composition of common chrome ores /mass%
| Source of chrome ore | South Africa | India |
|---|---|---|
| SiO2 | 0.31 | 0.83 |
| Al2O3 | 14.66 | 10.54 |
| Fe2O3 | 27.87 | 16.71 |
| CaO | 0 | 0.36 |
| MgO | 10.28 | 12.67 |
| Cr2O3 | 46.12 | 58.71 |
| LOI | 0.76 | 0.18 |
| Compounds in chrome ore | Formula | Iron valence | S-Q of South Africa | S-Q of India |
|---|---|---|---|---|
| Chromite, magnesian | (Fe0.51Mg0.49)(Cr0.73Al0.27)2O4 | Fe2+ | 34.8% | 31.6% |
| Iron silicon spinel | (Fe0.324Si0.676)(Fe0.963Si0.037)2O4 | Fe2+ + Fe3+ | 1.6% | 5.2% |
| Magnesium aluminum chromium spinel | (Al0.32Cr1.68)MgO4 | 3.2% | 16.8% | |
| Iron alumochromate | Fe(AlCrO4) | Fe2+ | 10.9% | 20.6% |
| Iron chromium trioxide | FeCrO3 | Fe3+ | 11.4% | |
| Magnesium aluminum iron spinel | MgAl0.8Fe1.2O4 | Fe3+ | 7.2% | 23.3% |
| Magnesioferrite, aluminian | (Mg0.32Fe0.68)(Al0.70Mg0.68Fe0.62)O4 | Fe2+ + Fe3+ | 22.5% | |
| Maghemite-Q, syn | Fe2O3 | Fe3+ | 6.9% | |
| Gibbsite, syn | Al(OH)3 | 0.6% | 2.0% | |
| Chlorite-serpentine | (Mg,Al)6(Si,Al)4O10(OH)8 | 0.9% | 0.5% |
Table 3 XRD semi-quantitative S-Q compounds in chrome ores of South Africa and India
| Compounds in chrome ore | Formula | Iron valence | S-Q of South Africa | S-Q of India |
|---|---|---|---|---|
| Chromite, magnesian | (Fe0.51Mg0.49)(Cr0.73Al0.27)2O4 | Fe2+ | 34.8% | 31.6% |
| Iron silicon spinel | (Fe0.324Si0.676)(Fe0.963Si0.037)2O4 | Fe2+ + Fe3+ | 1.6% | 5.2% |
| Magnesium aluminum chromium spinel | (Al0.32Cr1.68)MgO4 | 3.2% | 16.8% | |
| Iron alumochromate | Fe(AlCrO4) | Fe2+ | 10.9% | 20.6% |
| Iron chromium trioxide | FeCrO3 | Fe3+ | 11.4% | |
| Magnesium aluminum iron spinel | MgAl0.8Fe1.2O4 | Fe3+ | 7.2% | 23.3% |
| Magnesioferrite, aluminian | (Mg0.32Fe0.68)(Al0.70Mg0.68Fe0.62)O4 | Fe2+ + Fe3+ | 22.5% | |
| Maghemite-Q, syn | Fe2O3 | Fe3+ | 6.9% | |
| Gibbsite, syn | Al(OH)3 | 0.6% | 2.0% | |
| Chlorite-serpentine | (Mg,Al)6(Si,Al)4O10(OH)8 | 0.9% | 0.5% |
| Firing atmosphere from 800 °C to 1 650 °C | 3.0% to 5.6% O2 content (Fuel to air ratio < 1:1) | 0.5% O2 content (Fuel to air ratio > 1:1) | |||
|---|---|---|---|---|---|
| Grade of magnesia-chrome compaction | Low | High | Low | High | |
| Unfired density /(g · cm-3) | 3.08 | 3.22 | 3.08 | 3.23 | |
| Fired density /(g · cm-3) | 2.95 | 3.09 | 3.03 | 3.20 | |
| Firing expansion rate /% | 1.40 | 0.73 | 0.47 | 0.33 | |
| Apparent porosity /% | 19.5 | 18.0 | 16.5 | 16.0 | |
| Hot module of rupture /MPa | at 1 260 °C | 9.7 | 10.5 | 20.0 | 14.6 |
| at 1 482 °C | -- | 1.0 | 2.4 | 2.3 | |
Table 4 Influence of firing atmosphere on physical properties of magnesia-chrome brick
| Firing atmosphere from 800 °C to 1 650 °C | 3.0% to 5.6% O2 content (Fuel to air ratio < 1:1) | 0.5% O2 content (Fuel to air ratio > 1:1) | |||
|---|---|---|---|---|---|
| Grade of magnesia-chrome compaction | Low | High | Low | High | |
| Unfired density /(g · cm-3) | 3.08 | 3.22 | 3.08 | 3.23 | |
| Fired density /(g · cm-3) | 2.95 | 3.09 | 3.03 | 3.20 | |
| Firing expansion rate /% | 1.40 | 0.73 | 0.47 | 0.33 | |
| Apparent porosity /% | 19.5 | 18.0 | 16.5 | 16.0 | |
| Hot module of rupture /MPa | at 1 260 °C | 9.7 | 10.5 | 20.0 | 14.6 |
| at 1 482 °C | -- | 1.0 | 2.4 | 2.3 | |
| [1] |
Toshihiko EMI. Steelmaking technology for the last 100 years: Toward highly efficient mass production systems for high quality steels. ISIJ International, 2015, 55(1):36-66.
DOI URL |
| [2] | Dieter Tembergen, Rainer Teworte, Robert Robey. RH metallurgy. Millennium Steel 2008: 104-108. |
| [3] | Bill Foote, Anke Teeuwsen, Peter Raynerd. Ruhrstahl-Heraeus process with mechanical vacuum pumps: absurdity or red-hot technology and money well spent? Millennium Steel 2021: 66-71. |
| [4] |
Guangwei Yang, Xinhua Wang, Fuxiang Huang, Wanjun Wang, Yuqun Yin. Transient inclusion evolution during RH degassing. Steel Research International, 2014, 85(1):26-34.
DOI URL |
| [5] |
Dianqiao Geng, Jinxing Zheng, Kai Wang, Ping Wang, Ruquan Liang, Haitao Liu, Hong Lei, Jicheng He. Simulation on decarburization and inclusion removal process in the Ruhrstahl-Heraeus (RH) process with ladle bottom blowing. Metallurgical and Materials Transactions B, 2015, 46(3):1484-1493.
DOI URL |
| [6] |
Mun-Kyu Cho, Marie-Aline Van Ende, Tae-Hee Eun, In-Ho Jung. Investigation of slag-refractory interactions for the Ruhrstahl-Heraeus (RH) vacuum degassing process in steelmaking. Journal of the European Ceramic Society, 2012, 32(8):1503-1517.
DOI URL |
| [7] |
Dong Hyun Kim, Suk Hyun Yoo, Chang Soo Ha, Jong Min Park, Kyung Sub Lee, Sung Man Kim. Wear of direct bonded magnesia-chrome ore refractories in the RH-TOB process. Journal of the Ceramic Society of Japan, 2005, 113(6):405-408.
DOI URL |
| [8] |
M. Guo, P. T. Jones, S. Parada, E. Boydens, J. V. Dyck, B. Blanpain, P. Wollants. Degradation mechanisms of magnesia-chromite refractories by high-alumina stainless steel slags under vacuum conditions. Journal of the European Ceramic Society, 2006, 26(16):3831-3843.
DOI URL |
| [9] |
Zhangfu Yuan, Wenlai Huang, Kusuhiro Mukai. Local corrosion of magnesia-chrome refractories driven by Marangoni convection at the slag-metal interface. Journal of Colloid and Interface Science, 2002, 253(1):211-216.
PMID |
| [10] |
Gengfu Liu, Yawei Li, Tianbin Zhu, Yibiao Xu, Jun Liu, Shaobai Sang, Quanyou Li, Yuanjin Li. Influence of the atmosphere on the mechanical properties and slag resistance of magnesia-chrome bricks. Ceramics International, 2020, 46(8A):11225-11231.
DOI URL |
| [11] | Subramaniam Kumar, Alfred Kremer. Latest developments in RH degasser snorkels. Proceedings of UNITECR 2015, Vienna, Austria, 2015: No. 63. |
| [12] | Allen M. Alper. High Temperature Oxides, Part I: Magnesia, Lime and Chrome Refractories. Academic Press, New York, NY. 1971: 110-133. |
| [13] | Charles A. Schacht. Refractories Handbook. Marcel Dekker, Inc. New York, 2004: 109-149. |
| [14] |
Vilas D. Tathavakar, M. P. Antony, Animesh Jha. The physical chemistry of thermal decomposition of South African chromite minerals. Metallurgical and Materials Transactions B, 2005, 36:75-84.
DOI URL |
| [15] | Harbison-Walker Refractories Co. Handbook of Refractory Practice. Moon Township, PA. USA. 2008. CR-3. |
| [16] |
I. de Menezes, V. S. Stubican. Grain-boundary reactions in magnesia-chrome refractories: application of the electron probe, II. Journal of the American Ceramic Society, 1966, 49(11):609-612.
DOI URL |
| [17] | Zongqi Guo, Ying Ma. Bonding mechanisms of basic bricks for RH snorkels. China’s Refractories. 2020, 29(4):29-34. |
| [18] | H. Barthel, F. Kassegger, F. Soucek. The use of magnesia chromite refractories made from sintered co-clinker in secondary steelmaking and open-hearth furnace. Proceedings of International Symposium on Refractories, Hangzhou, China, 1988: 489-504. |
| [19] | Long Li, Jingkun Yu, Zongshu Zou. Reaction between high grade magnesia and Indian chrome ore during firing. China’s Refractories, 2007, 16(s):155-157. |
| [20] |
Rui Li, Yong Li, Dong Wang, Yang Li. The influence of Indian concentrate chrome ore on top quality magnesite-chrome brick for RH degasser. Advanced Materials Research, 2011, 233-235:2654-2659.
DOI URL |
| [21] |
Kiyoshi Gotot, William E. Lee. The “direct bond” in magnesia chromite and magnesia spinel refractories. Journal of the American Ceramic Society, 1995, 78(7):1753-1760.
DOI URL |
| [22] | Kenji Ichikawa, Ryosuke Nakamura, Masanori Ogata, Minoru Suto. Distribution and properties of secondary spinel in magnesia-chrome bricks. Shinagawa Technical Report, 1996, 39:25-34. |
| [23] | Grand M. Farrington, Walter S. Treffner. Direct bonded refractory bricks. United States Patent, 3829541. 1972-04-19. |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||